Pasteur Institute – Laboratories
Laboratories Athens 2022 Design & Construction of 100 sq.m high risk Biosafety Laboratories BSL 3, for the Microbiology Department of Pasteur Institute in Athens, Greece. The design is in accordance
From design to construction to maintenance, BCT Group has multiyear experience in delivering fully functional BSL and GMP labs, meeting the unique needs of each project with detailed planning and coordination in every step.
Biosafety Laboratories (BSL LABS) treat biological, pathogenic or toxic agents.
Design & Built of those spaces aims in assuring protection of the working personnel and the community of the pathogenic substances unintentionally released in the laboratory environment.
World Health Organization (WHO) divides BSL Labs into three categories regarding the biosafety Requirements: Basic (core) Control, Heightened Control, Maximum Heightened Control. Previous version 4 categories (BLS 1,2,3 & 4) determining the basic principles and requirements for each case.
BLS Labs certification follow the Standards of ABSA Association.
For BSL Labs we undertake the design, construction, integration of special HVAC equipment for the proper operation of the LAB, Laboratory Equipment, Security and ABSA Certification as turn-key solution.
BCT company has developed numerous BSL labs in Greece as well as the only BSL Class 3 in Athens, realized in Pasteur institute and certified by ABSA.
GMP laboratories are part of the QC activities in a Pharmaceutical / Medical Manufacturing suite or operate independently for R&D and prototyping purposes of new products.
GMP Labs need to comply with Good Manufacturing Practices Standards for this reason structural materials must be flash clean and shall not shed particles, Air filtration system must assure the required ISO 14644 cleanliness class according to GMP Class (A/B,C or D) and the necessary differential pressures. Equipment, lighting, laboratory gases circuits, security and monitoring are objectives that need to be taken care of.
In same Laboratories special engineering issues must be taken into account such as energy regain, redundancy as well as minimizing of energy consumption.
Our company designs and builds GMP Laboratories as a turn key solution providing IQ, OQ certification of the LAB to be submitted to the authorities for the acquisition of the necessary license.
We can also provide furniture and equipment so as the Laboratory to be read for use.

Laboratories Athens 2022 Design & Construction of 100 sq.m high risk Biosafety Laboratories BSL 3, for the Microbiology Department of Pasteur Institute in Athens, Greece. The design is in accordance
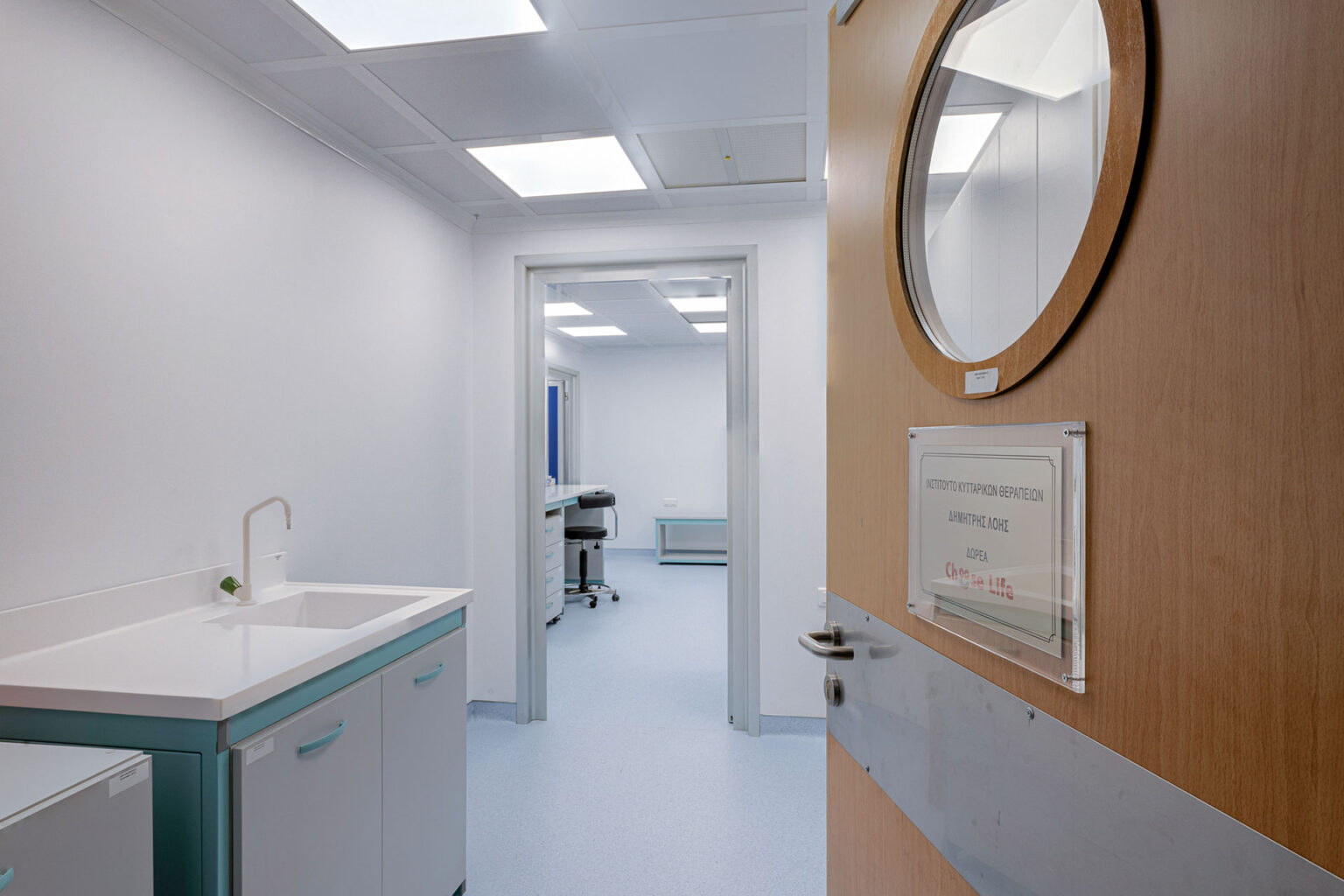
Laboratory Patra, Greece 2021 Design, Construction & Validation of a complex of Cleanrooms which are used as Laboratories to research innovative therapies for hematological cancer in the Medical School of

Cleanroom GMP Class A/B Re-Design & Construction for the Quality Control Department of the Hellenic Pasteur Institute. It is used as a laboratory for the quality control of injectables. Laboratories