Laboratory

Thrakomakedones, Greece

2020
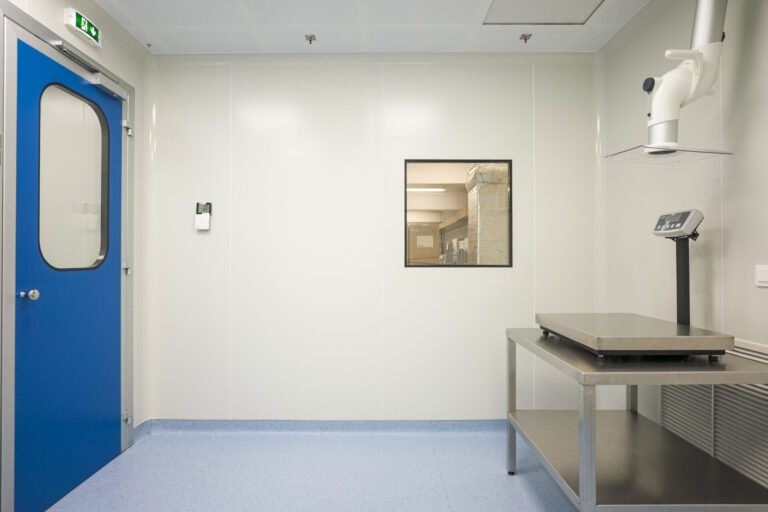
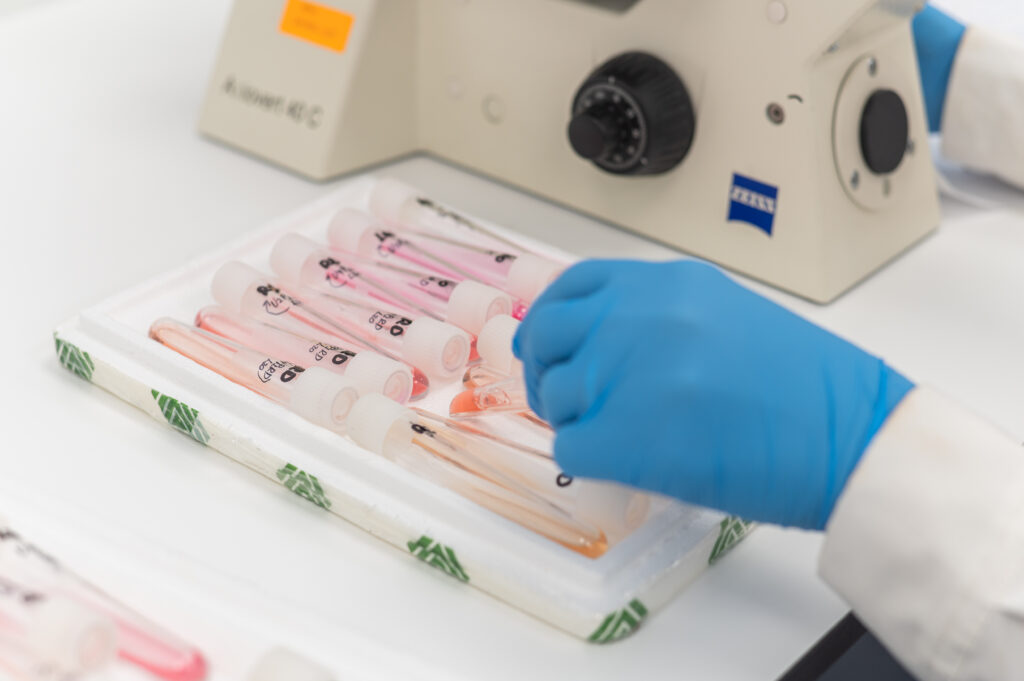
This project concerned the reconstruction of facilities in the National Hematology Center, to form a suite of 4 Biosafety Laboratories in which hematological analysis are performed in conditions to avoid contamination and infection spread. The laboratories serve as Covid-19 samples examination centers.
An EuGMP Class B laboratory set up in which the samples are opened for their microbiological control, two airlocks of B Class for personnel to enter and leave the cleanroom and a Class D space where the samples are received without coming into contact with the laboratory personnel.
All areas are under vacuum so that no air escapes from the laboratory to other areas outside.
Our company was entrusted in a very limited time (max 2 weeks) and in the conditions of quarantine due to the COVID-19 pandemic to complete this project and help deal with the pandemic.
We worked hard taking all the essential safety measurements, we used our team spirit, we took advantage of our high expertise and specialization, and we manage to accomplish 100m2 Laboratories within six days.
Commissioning and Certification according to ISO 14644 & EU GMP Volume 4 Annex 1 also accomplished by BCT specialists and the appropriate DQ, IQ and OQ protocols were provided upon completion of the process.